Navigating CEA Food Safety: Candid Q&A With Ceres Certifications, International President
Join us for a candid conversation with Dr. Karl Kolb, President of Ceres Certifications, International, and Ceres University, as he sheds light on the essentials of food safety in controlled environment agriculture (CEA). Ahead of his CEA Food Safety Workshop at the March 2024 edition of Indoor Ag-Con, Dr. Kolb delves into the practical aspects of GFSI (Global Food Safety Initiative) standards, addressing common misconceptions. From understanding the risk-based approach in CEA to incorporating technology into safety measures, this Q&A provides straightforward insights. Dr. Kolb also offers pragmatic advice on how CEA operators can balance sustainability with the need for robust food safety.
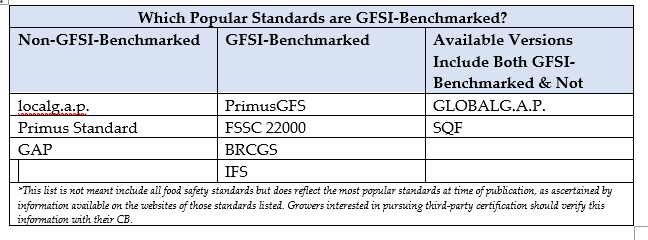
How does the application of GFSI standards benefit controlled environment agriculture (CEA) operations, and what specific challenges or considerations should CEA operators be aware of when seeking food safety certification for their facilities?
Let me start by saying GFSI food safety standards are largely misunderstood across the entire food industry. From their development, what they represent and how they are applied. It may sound unusual but if the owners and operators of food operations understood them, they would place them as their top priority in their daily schedule. The leaders would not go home at night nor would any of their employees until every item in their food safety plan was checked, doublechecked and rechecked.
 There are so many unique ways a GFSI or any “certificated” (non-GFSI) food safety standard benefits the operator. Defining “Operator” is anyone directly involved in any aspect of the food operation. From those who sell inputs such as seed, to those who plant the seed, those who irrigate, control watering, clean and sanitize, pull maintenance, process or pack, sell and ship the finished product — they are all operators.
There are so many unique ways a GFSI or any “certificated” (non-GFSI) food safety standard benefits the operator. Defining “Operator” is anyone directly involved in any aspect of the food operation. From those who sell inputs such as seed, to those who plant the seed, those who irrigate, control watering, clean and sanitize, pull maintenance, process or pack, sell and ship the finished product — they are all operators.
Literally the information on food safety programs and their attributes would fill the Library of Congress. And the quality systems that are used to apply the “standard” as we say, would fill a second Library of Congress. Let me be clear, the application of a GFSI or food safety standard is the same across the board, to any food safety operation, CEA or the larger food industry. That statement scares CEA operators. CEA operators, like organic growers or small farmers, spend their life differentiating themselves from their counterparts. Each one, and rightfully so believes and feels in their heart that they are uniquely different and performing the most important service to the public. I cannot disagree with any of this because each operator is doing a phenomenal service to the largely unknowing public.
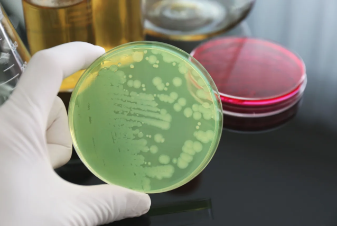
CEA operators should not fear the application of the GFSI standard. If, and that is a big if to be discussed later, it is applied correctly the CEA operator would intuitively know how greatly it benefits them. In short, each standard in a food safety scheme (Schemes are GFSI programs such as SQF, PGFS and HACCP to name a few.) is based on one of three or all three tenants; science, regulatory codes, and industry norms. While there are variations of the GFSI schemes to accommodate the differences in some farming activities such as greenhouse farming by example, largely all food safety schemes inherently possess the same core requirements. It’s how they are applied that makes the difference in each different operation.
Are there specialized considerations for food safety in CEA that may differ from traditional agriculture, and if so, how can operators navigate these nuances?
Herein is the one of my favorite topics about the application of the standard. It is risk based. In other words, each standard is applied the same but differently, based on a risk analysis. No matter what part of the food industry the operation is working in, the standard is the same. This fact alone is one of the particular strengths of the GFSI system. The “specialized considerations” mentioned in your question are invoked at the time the auditor asks the question from the standard. Navigating these nuances as you stated are not what most people believe – for sure it’s not fancy footwork during an audit or attractive paperwork. By example, I write some of the most boring, grammatically incorrect, ugly looking programs and policies, reports and logs ever. Pretty and poetic is great but it doesn’t necessarily mean there is a meaningful robust program, “under the hood”, so to speak.
 The “navigation” begins at the beginning (There is a song by Artie Shaw with a similar name referring to a dance or relationship from the 1920s that may have some carryover when applying the standard?) and thrives throughout the program and over time. It is the analysis of the particular “clause” in a food safety standard – this analysis begins with a complete understanding of what the clause is asking specifically and how it is intended to be applied. Remember I spoke earlier about how a standard or clause is built? Science, regulatory and industry norms? A short explanation of Risk Analysis is in order to understand the next piece. A risk analysis is a process which entails identifying risk, defining uncertainty, completing analysis models and implementing solutions.
The “navigation” begins at the beginning (There is a song by Artie Shaw with a similar name referring to a dance or relationship from the 1920s that may have some carryover when applying the standard?) and thrives throughout the program and over time. It is the analysis of the particular “clause” in a food safety standard – this analysis begins with a complete understanding of what the clause is asking specifically and how it is intended to be applied. Remember I spoke earlier about how a standard or clause is built? Science, regulatory and industry norms? A short explanation of Risk Analysis is in order to understand the next piece. A risk analysis is a process which entails identifying risk, defining uncertainty, completing analysis models and implementing solutions.
Now I must back up a bit. Life is about backing up and moving forward. Just like learning from an audit. The risk analysis is where the CEA operator differs from the larger industry and even his co-operators down the street. There is a yin and yang relationship between the auditor and the operator. The auditor applies a process involving the standard or clause. The auditor understands the standard and clause. He looks to the operator as the expert on the ground to explain how the operator applied the principles of risk analysis to the standard and what the operator’s solution or program properly answers the clause.
I’m sure by now the questions of how CEA differs from the larger industry and its fellow brethren are becoming clear.
Two absolutes must be respected in this dance (somehow credit Artie) or the yin and yang relationship. 1) The auditor must know his job and understand his or her role in the audit, 2) The operator must know his or her job of performing a risk analysis and explaining it to the auditor. That is the strength and difference CEA operators are looking for in GFSI audits. However, finding #1 and #2 is like finding “hen’s teeth”. So sadly, audits become a checklist affair, almost worthless and get a bad rap.
As a leader of a food safety certifying body, you’ve likely encountered various compliance issues. What are some common misconceptions or overlooked aspects related to food safety that you believe CEA operators should be more aware of to improve the safety of their produce?
Not sure if I’m a leader but more of a learner or supporter. We are largely a body of awesome women who I absolutely (adore) believe are the strength of this organization. I have chosen smart, educated, dedicated and loyal women who don’t need to be told what to do or how to do it.
Our challenge is many-fold. It involves resources. Time, money, training, staff needs and lastly, but first, compliance.
 My academic background is about quality. Quality is defined properly, partially by the ISO system and my experience, “Quality standards are sets of good manufacturing practices (“Best Practices”), methods, systems, requirements, and or specifications established by science, regulators and industry to help operators achieve and demonstrate consistent production and product qualities.” Do not confuse quality with quality. We are not talking about quality like the organoleptic head of lettuce qualities, although quality systems do define this commodity standard. Quality is all about consistency.
My academic background is about quality. Quality is defined properly, partially by the ISO system and my experience, “Quality standards are sets of good manufacturing practices (“Best Practices”), methods, systems, requirements, and or specifications established by science, regulators and industry to help operators achieve and demonstrate consistent production and product qualities.” Do not confuse quality with quality. We are not talking about quality like the organoleptic head of lettuce qualities, although quality systems do define this commodity standard. Quality is all about consistency.
A great and successful example is McDonalds. Sadly, my default menu on too many occasions. They grew fast and successfully by using a quality model. The bros McDonald correctly set their goal as fast, good, cheap and consistent burgers across the land. It’s not that the burgers are the best ever (sorry bros however the fish sandwich is the best) but everything from the sandwich itself to the service is consistently the same. Go anywhere and the McDonalds experience is not 100% every time, listen well, it’s the same experience every time. Manufacturing excellence is achieved through consistency. And to those who are manufacturers we know that it’s not 100% that is achieved every time but the 90% mark is where quality is achieved.
Compliance at the operator level is all about consistency. A food safety program cannot run at 100%, but it can run properly at 90% and achieve science, regulatory and industry expectations. The challenge of both myself as a certification body and that of the operator is keeping up and applying the science, regulations and industry expectations in auditors and operators as they work though (think root cause analysis) risk analysis solutions.
The audit is not meant to be a checklist drill but the yin and yang of auditor and operator.
As technology continues to play a significant role in CEA, how do advancements in automation and data-driven systems impact food safety protocols, and what advice do you have for growers looking to integrate these technologies while maintaining a strong food safety program?
Automation should serve the food safety program, not drive, define or prescribe it. I once asked a very wise and experienced individual with a very large certifying body how he conducted audits. This gentleman told me he would take a blank yellow pad and walk into a food plant and begin asking questions. He had infinite knowledge of the standard. He went and asked questions until he got the answers that rang true. This is the ultimate in determining the robustness of a food safety program. I’m sure the yellow pad had a lot to do with it too.
As I taught in the classroom, you define automation, don’t let it define you. All too often we fall for the “sizzle” of what these systems are said to do and we find out the sizzle is not from a tenderloin but a burger. (My apologies to the bros McDonald.)
With the growing importance of sustainability in agriculture, how can CEA operators balance the use of sustainable practices with the need for rigorous food safety measures. Are there specific certifications or guidelines they should consider?
 As you can tell by now, my perspective on food safety is different from anything else – I learned as a manufacturing engineer that when things don’t work as they should (different from the standard) you go back to the basics and start over – in this case, the basics of quality.
As you can tell by now, my perspective on food safety is different from anything else – I learned as a manufacturing engineer that when things don’t work as they should (different from the standard) you go back to the basics and start over – in this case, the basics of quality.
I started this interview by saying GFSI food safety standards are largely misunderstood across the entire food industry. Here again, I must separate the norm from what I believe is important. We as an industry do not understand quality systems and their concepts or requirements.
Sustainability is all about quality systems. Quality systems are not a point in time like an audit. Nor is sustainability. The question is, “How do we sustain quality?” Sustainability has become defined as a social construct. Wrong. Sustainability is doing the same thing consistently and improving time after time for a sustained period of time. Not more or new twists of the standard.
I have tried in this interview to impress your readers that the GFSI system must be embraced in a quality fashion. As a process and not in a one-time checklist inspection. While we gloat that GFSI is the best food safety system in the world and the US leads the way, we all drank the kool-aid.
We have been lucky as a nation illness-wise, to date. As the demand for food increases what we do now, what we call food safety of trying to pass a once-a-year chaotic intervention (annual audit) of our operations, is not sustainable.
A good friend (Bob Wright) sums it this way, “Does it make the food any safer?”
Thanks for listening and apologies to anyone offended, especially the bros McDonald.
 Karl Kolb, Ph.D., is the founder and President of the High Sierra Group companies, which services more than 10,000 customers with Ceres Certifications, International (ISO 17065 food safety certifying body), HSG/AME Certified Laboratories (17025 food testing laboratories), Ceres University (Accredited, degree granting), High Sierra Chemicals and Epicure Farms.
Karl Kolb, Ph.D., is the founder and President of the High Sierra Group companies, which services more than 10,000 customers with Ceres Certifications, International (ISO 17065 food safety certifying body), HSG/AME Certified Laboratories (17025 food testing laboratories), Ceres University (Accredited, degree granting), High Sierra Chemicals and Epicure Farms.
Unlock the Secrets To A Safer, Higher Quality Harvest With March 2024 CEA Food Safety Workshop Registration Fee Includes
Expo Floor Access & Up To 3 CEUs
Internal auditing certification is a mandatory GFSI (Global Food Safety Initiative) requirement that demonstrates an individual’s ability to conduct internal assessments of any food safety program. Indoor Ag-Con has partnered with Ceres University, a leading provider of IACET-accredited food safety training and certification, to offer a cost-effective, convenient way to build your career AND help fulfill GFSI scheme requirements. Workshop fee includes:
- Admission to 4-hour workshop and course materials
- Ability to earn up to 3 Continuing Education Units (CEUs) upon completion
- Indoor Ag-Con Expo Hall Only Pass, which includes access to Expo Floor March 11-12, 2024; admission to all Indoor Ag-Con Expo Theater presentations; Expo Floor Welcome Happy Hour; and access to expo floor of National Grocers Association (NGA) Show running concurrently at Caesars Forum.








 Workshop instructors include Dr. Karl Kolb, president of Ceres University and Ceres Certifications, International (CCI) and Kellie Worrell, GlobalG.A.P. Scheme Manager, CCI. Dr. Kolb is a microbiologist with a quality background and more than 30 years as an industry professional. In addition to her current role with CCI, Kellie Worrell has managed the Food Safety Program for multiple vegetable farms, including a wide variety of crops. CCI features GLOBALG.A.P. among its many GFSI food safety schemes.
Workshop instructors include Dr. Karl Kolb, president of Ceres University and Ceres Certifications, International (CCI) and Kellie Worrell, GlobalG.A.P. Scheme Manager, CCI. Dr. Kolb is a microbiologist with a quality background and more than 30 years as an industry professional. In addition to her current role with CCI, Kellie Worrell has managed the Food Safety Program for multiple vegetable farms, including a wide variety of crops. CCI features GLOBALG.A.P. among its many GFSI food safety schemes.

 About Ceres Certifications, International
About Ceres Certifications, International
 About Ceres Certifications, International
About Ceres Certifications, International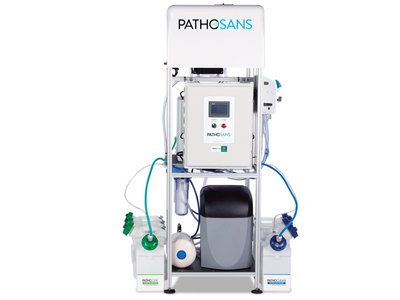
 Delivering “Greenhouse-as-a-Service” (GaaS), EGT designs, engineers and manufactures vertical greenhouses to help grow large amounts of fresh food that can be sourced locally. The company partnered with PathoSans because unlike traditional caustic cleaning chemicals, PathoSans’ ECAS technology produces a cleaner and disinfectant using just salt, water and electricity. It contains no added fragrances and is safe for use on foods and in food preparation. In addition, the disinfectant eliminates pathogens and molds that can exist on food, where food grows, and in food packaging and preparation areas.
Delivering “Greenhouse-as-a-Service” (GaaS), EGT designs, engineers and manufactures vertical greenhouses to help grow large amounts of fresh food that can be sourced locally. The company partnered with PathoSans because unlike traditional caustic cleaning chemicals, PathoSans’ ECAS technology produces a cleaner and disinfectant using just salt, water and electricity. It contains no added fragrances and is safe for use on foods and in food preparation. In addition, the disinfectant eliminates pathogens and molds that can exist on food, where food grows, and in food packaging and preparation areas. “Food safety is our top priority at EGT and it governs each of our processes,” said Aaron Fields, Director of Horticulture at Eden Green Technology Fields. “That’s why we chose PathoSans to ensure our operations use safe and effective cleaning solutions. PathoSans shares our mission of delivering innovative technology with a focus on social responsibility, safety, self-sufficiency, and sustainability.”
“Food safety is our top priority at EGT and it governs each of our processes,” said Aaron Fields, Director of Horticulture at Eden Green Technology Fields. “That’s why we chose PathoSans to ensure our operations use safe and effective cleaning solutions. PathoSans shares our mission of delivering innovative technology with a focus on social responsibility, safety, self-sufficiency, and sustainability.” Through its partnership with PathoSans, EGT can also fulfill its mission of providing greenhouses and services that reduce waste, water consumption and reliance on greenhouse gas emissions. On-site generation of cleaning and disinfecting solutions allows EGT and its clients to refill spray bottles and reduce plastic waste. Without relying on chemical supply chains, EGT further shrinks its carbon footprint by eliminating chemical transportation, storage and disposal. Additionally, since PathoSans solutions leave no chemical residue behind on surfaces, greenhouses can save water since less rinsing is required.
Through its partnership with PathoSans, EGT can also fulfill its mission of providing greenhouses and services that reduce waste, water consumption and reliance on greenhouse gas emissions. On-site generation of cleaning and disinfecting solutions allows EGT and its clients to refill spray bottles and reduce plastic waste. Without relying on chemical supply chains, EGT further shrinks its carbon footprint by eliminating chemical transportation, storage and disposal. Additionally, since PathoSans solutions leave no chemical residue behind on surfaces, greenhouses can save water since less rinsing is required.
 What started in 2011 as a dream to deliver fresh, nutritious produce to Canadians year-round has become reality. As Canada’s first and largest commercial vertical farming operation,
What started in 2011 as a dream to deliver fresh, nutritious produce to Canadians year-round has become reality. As Canada’s first and largest commercial vertical farming operation,  We are currently growing microgreens — Spicy Mustard Medley, Pea Shoots, Asian Blend, Micro Arugula, Micro Radish, and Micro Broccoli — and baby greens – Ontario Arugula, Ontario Spring Mix and Ontario Baby Spinach.
We are currently growing microgreens — Spicy Mustard Medley, Pea Shoots, Asian Blend, Micro Arugula, Micro Radish, and Micro Broccoli — and baby greens – Ontario Arugula, Ontario Spring Mix and Ontario Baby Spinach. Our sustainable practices include:
Our sustainable practices include: Construction on our Calgary farm is well under way. We are on schedule to have the 95,000-square-foot farm open and providing fresh leafy greens to grocery stores across Western Canada in first half of next year.
Construction on our Calgary farm is well under way. We are on schedule to have the 95,000-square-foot farm open and providing fresh leafy greens to grocery stores across Western Canada in first half of next year. We are constantly innovating and experimenting with new processes and products to bring the best possible leafy greens to Canadian consumers.
We are constantly innovating and experimenting with new processes and products to bring the best possible leafy greens to Canadian consumers.

